- It can be made almost anywhere, so a worldwide trade in the net-zero era will never rival the oil industry.

By David Flickling
If you want a symbol of how energy is a global industry as fundamental as the trades in metals or government bonds, one image has held sway for decades: The monumental black-and-red hull of a crude oil supertanker.
It’s only natural, then, that a world transitioning to cleaner sources of energy should seek out a comparable emblem for the net-zero era. A prime candidate to replace petroleum is another substance that can be moved around in tankers: green hydrogen (so-called because it’s produced by using renewable energy to split apart water molecules. 1 )

Some 400 million metric tons of hydrogen a year (not necessarily green) will be moved over long distances by 2050, according to the Hydrogen Council, which represents the nascent industry. Countries rich in cheap renewables, such as Australia, Brazil and Chile, hope to become H2 hubs every bit as pivotal to the global economy as the Persian Gulf is in our current hydrocarbon era. Even Saudi Arabia is working on a $8.5 billion green hydrogen plant.
Will a worldwide trade in hydrogen grow to take on the same role that the oil industry has right now? It’s not likely. To see why, it’s worth looking at how green hydrogen will be made, transported and used — and considering the parallels with existing flows of commodities.
One binding constraint on commerce since the dawn of history has been the cost of transport. Only higher-value items are worth moving over long distances. Ancient China and Rome traded silk and glassware, but not wheat and rice. It’s not so different these days. You’ll typically pay between $10 and $50 a ton to ship a non-containerized product like oil, wheat or coal across an ocean. As a result, materials like cement and sulfur — abundant and relatively cheap — are mostly consumed close to where they’re produced. It’s simply not worthwhile putting them on a ship.

For most commodities, trade rises with increasing prices. The exceptions to that rule are telling, as the above chart illustrates. High-quality reserves of iron ore and crude oil are scarce globally, so consumers have no choice but to transport them from further afield. Technological innovation will never make Japan a major petroleum producer, or South Korea a powerhouse of iron ore. Geology is destiny, giving those commodities a special cachet.
Green H2 is different. No major economy faces a permanent shortage of hydrogen’s raw materials: water and renewable energy. In that sense, it more closely resembles products like gypsum and ammonia, which can be manufactured almost anywhere.
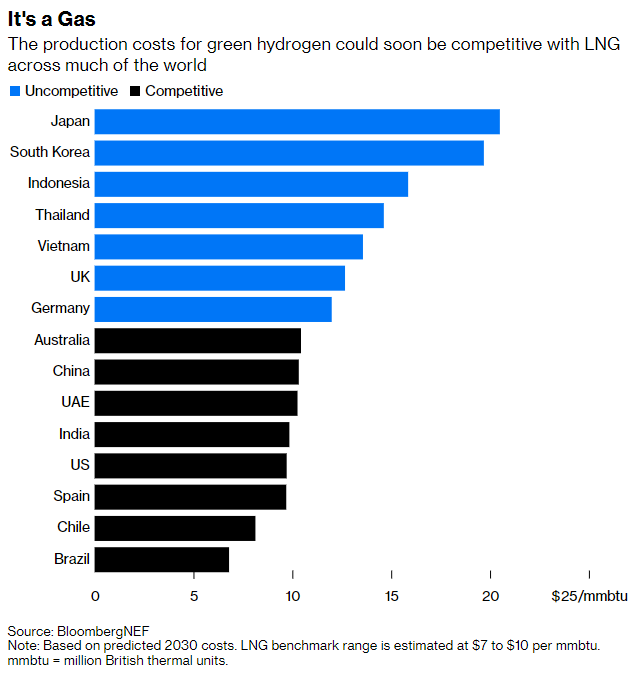
The prices of renewable energy do differ from country to country, to be sure — but not by enough to overcome the transport issue. Hydrogen is hard to move in its raw form, being highly reactive, far less dense than liquefied natural gas, and very, very cold. It only liquefies at minus 253 degrees Celsius (minus 423 Fahrenheit), almost as far below the temperature of LNG as ice is from steam. The costs of chilling substances increase dramatically the colder they get.
Most plans to solve this issue involve putting the hydrogen through a reactor at either end of the journey to convert it into a more easily-transportable form, with the prime candidates being ammonia, methanol, and the toluene used in paint thinner. The amount of energy used to drive those reactions, however, pushes the costs up further. Because of that, even Brazil — with some of the cheapest renewables — will struggle to build an export trade that can compete with domestically-produced green hydrogen.

Note: Based on hydrogen conversion via ammonia. Assumed hydrogen production cost of $1 per kilogram. Converted to mmbtu at a rate of 7.44kg/mmbtu.
You can shave these costs somewhat if you burn ammonia directly as fuel rather than attempting to convert it back to hydrogen, but the challenges involved have thwarted engineers since World War II, and chemists are still getting to grips with the processes involved. Burning ammonia is also a potent source of NOx particulates, one of a suite of major pollutants responsible for about 6.7 million deaths a year. It even generates nitrous oxide, a chemical that warms the atmosphere about 273 times as much as carbon dioxide that is rarely produced by current industrial activities.
Green hydrogen may yet remake the world of energy. In many parts of the world, it will be well within range of the $7 to $10 per million British thermal units cost of natural gas by the end of this decade, according to BloombergNEF. That should enable it to displace fossil fuels in a swath of applications requiring high-temperature heat, large-scale energy storage, or molecules for chemical compounds.
What it won’t do, however, is generate fleets of tankers connecting the globe. Like sulfur and ammonia — and the existing hydrogen trade that consumes nearly 100 million tons a year — it’s going to be used close to the place it was produced. Many countries may aspire to become the Saudi Arabia of the green hydrogen era. None will achieve that dream.
____________________________________________________________
David Fickling is a Bloomberg Opinion columnist covering commodities, as well as industrial and consumer companies. He has been a reporter for Bloomberg News, Dow Jones, the Wall Street Journal, the Financial Times and the Guardian. Energiesnet.com does not necessarily share these views.
Editor’s Note: This article was originally published by Bloomberg Opinion, on May 16, 2023. All comments posted and published on EnergiesNet.com, do not reflect either for or against the opinion expressed in the comment as an endorsement of EnergiesNet.com or Petroleumworld.
Use Notice: This site contains copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available in our efforts to advance understanding of issues of environmental and humanitarian significance. We believe this constitutes a ‘fair use’ of any such copyrighted material as provided for in section 107 of the US Copyright Law. In accordance with Title 17 U.S.C. Section 107. For more information go to: http://www.law.cornell.edu/uscode/17/107.shtml.
bloomberg.com 05 17 2023